PHDP5 Research for Alzheimer Disease
Introduction
Currently, there is no cure for Alzheimer’s disease, a progressive neurodegenerative condition that affects millions of people worldwide. However, treatments are available to help manage its symptoms, such as memory loss, confusion, and difficulties with thinking and reasoning. While these therapies do not stop the disease, some have shown promise in slowing its progression, offering hope for improving the quality of life for those affected.
Alzheimer’s is characterized by the abnormal accumulation of proteins in the brain, such as beta-amyloid plaques and tau tangles, which interfere with the transmission of nerve impulses and disrupt normal brain function. These proteins build ups damage and ultimately kill brain cells, leading to the cognitive and functional decline associated with the disease.
In a groundbreaking study, researchers tested a synthetic peptide on mice genetically engineered to develop Alzheimer’s-like symptoms. The findings were promising, showing that the treatment significantly reduced the buildup of these harmful proteins in the brain. Even more compelling, the peptide therapy appeared to restore key cognitive functions, such as memory and learning, which are typically impaired in Alzheimer’s patients.
This study written about below highlights the potential of innovative therapeutic approaches to target the root causes of Alzheimer’s, rather than just addressing its symptoms. While further research, including human trials, is necessary to validate these findings, this development represents a significant step forward in the quest for more effective treatments and, ultimately, a cure for Alzheimer’s disease.
The problem
As global life expectancy continues to rise, dementia has become an increasingly pressing public health challenge. The prevalence of dementia is projected to grow significantly, with studies estimating that over 150 million people worldwide could be affected by 2050. This staggering statistic underscores the urgent need for effective prevention and treatment strategies.
Alzheimer’s disease, the most common form of dementia, is characterized by a variety of debilitating symptoms, including memory loss, cognitive decline, and personality changes. These symptoms are widely believed to result from the accumulation of two key proteins in the brain: beta-amyloid (Aβ) and tau. Beta-amyloid forms sticky plaques between neurons, while tau creates tangles within neurons, disrupting their structure and function. Together, these protein abnormalities lead to widespread neuronal damage, brain shrinkage, and the hallmark symptoms of Alzheimer’s.
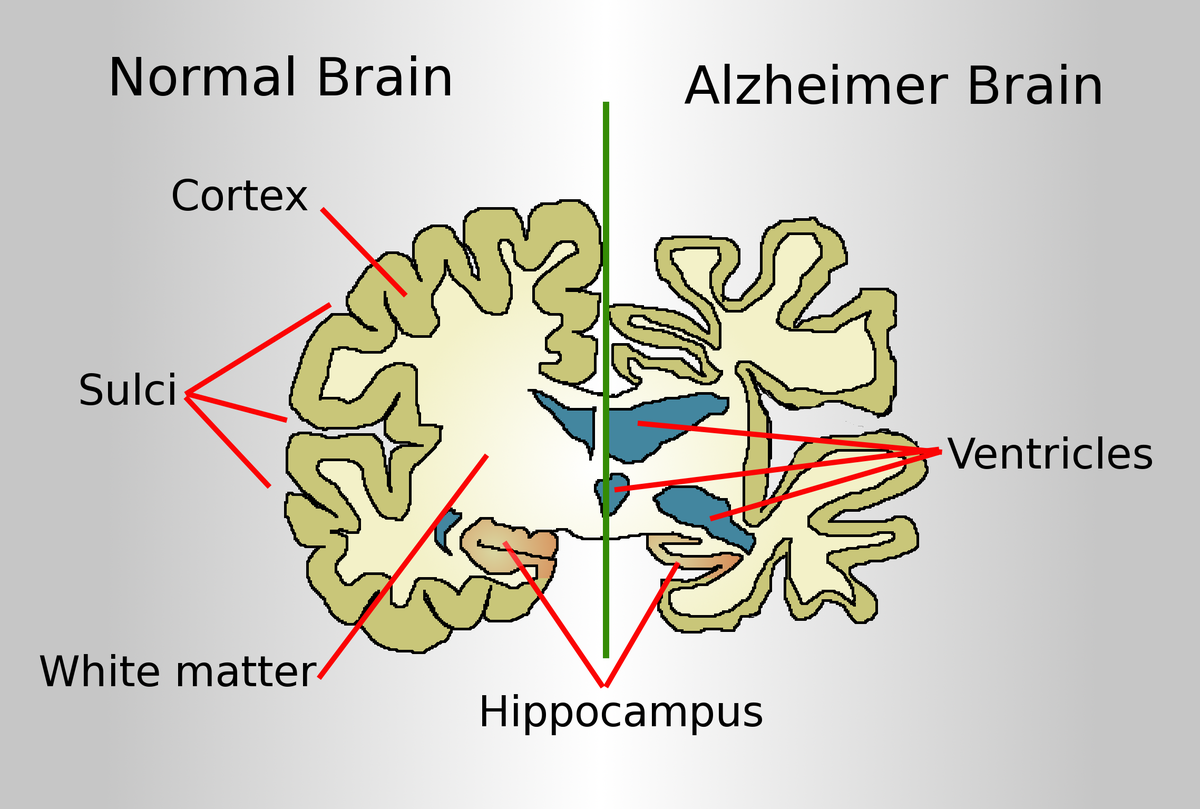
Current treatments for Alzheimer’s primarily focus on alleviating symptoms, such as cognitive deficits and behavioral changes, rather than addressing the underlying causes of the disease. However, recent advancements have introduced disease-modifying therapies that aim to directly target these pathological proteins. For example, aducanumab and lecanemab, two monoclonal antibody treatments, have shown promise in reducing Aβ plaques in the brain. These therapies mark a significant step forward in targeting the disease at its root.
Despite these advancements, the use of monoclonal antibody treatments has sparked debate within the medical community. While they offer